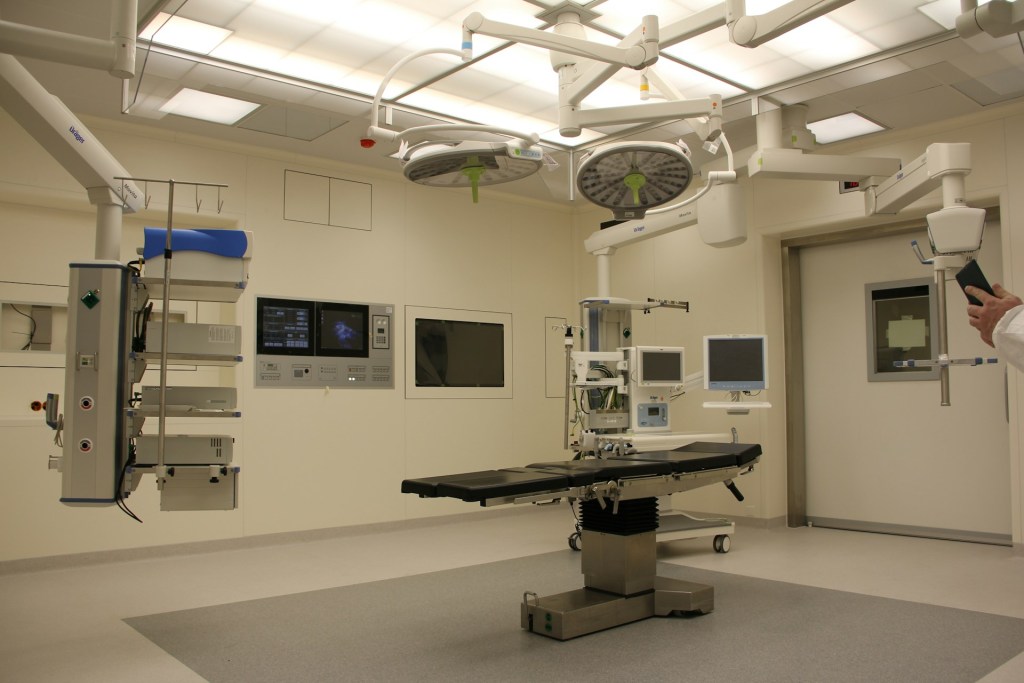
enBio ComplianceConnect
enBio ComplianceConnect provides medical device compliance consulting services to help hospitals meet Joint Commission and CMS requirements with confidence.

Regulatory Compliance Consulting for Hospitals and Clinics
Healthcare compliance isn’t just about passing audits; it’s about protecting patients, improving outcomes, and staying ahead of regulatory challenges. Our biomedical engineering consulting team works closely with your staff to integrate best practices in medical device safety, testing, and documentation. enBio ComplianceConnect was created to help hospitals and clinics meet the evolving demands of CMS and accrediting bodies such as The Joint Commission (TJC), DNV, HFAP, and CIHQ through clear, actionable support and medical device compliance consulting services.
As one of the trusted biomedical engineering consulting firms, we deliver hospital Joint Commission consulting support that focuses on both immediate readiness and long-term compliance.
Our team works alongside your leadership, clinical engineering, and facilities departments to ensure you’re fully prepared for survey readiness, with no surprises.
We Support Compliance In

CMS and Joint Commission audit preparation

Accreditation readiness for DNV, HFAP, and CIHQ

Documentation review and corrective action planning

Gap assessments and procedure alignment

Medical staff readiness and mock survey support
Consulting to maintain year-round compliance
Backed by enBio’s operational and technical expertise, ComplianceConnect bridges the gap between day-to-day operations and long-term regulatory compliance consulting strategy. We focus on high-risk areas that are often overlooked, helping you build a culture of readiness, not just check boxes.
Learn more in our ComplianceConnect press release
Guided by Proven Hospital Leadership
ComplianceConnect benefits from the insight of seasoned hospital leaders, including Lloyd Duplechan, former COO of major health systems and a recognized expert in hospital operations and accreditation readiness. His experience strengthens our biomedical consulting approach, helping us deliver practical hospital joint commission consulting and cms compliance consulting solutions that fit the real-world challenges healthcare facilities manage every day. We also bring expertise in hospital Joint Commission consulting and CMS compliance consulting to support continuous readiness and regulatory success.
How Our Biomedical Consulting Process Works
At enBio, we follow a structured approach to help healthcare facilities meet Joint Commission standards and CMS requirements. Our specialists in regulatory compliance consulting for medical devices guide hospitals through every step, from initial assessment to full accreditation readiness. Here’s how our biomedical consulting process works from start to finish.
